Anand Classes explains the atomic radius of elements shows a clear trend when we move down a group in the periodic table. For both alkali metals (Group 1) and halogens (Group 17), the atomic radius increases as we go from top to bottom. This happens because each successive element down the group has an additional electron shell in its electronic configuration. The added shell increases the distance between the outermost electrons and the nucleus, while the shielding effect of inner electrons reduces the effective nuclear pull. As a result, the atoms become larger despite the increasing nuclear charge.
Table of Contents
Variation of Atomic Radius in a Group
When we move down a group in the periodic table, the atomic radius of elements increases from top to bottom.
Reason for Increase in Atomic Radius
- Increase in Nuclear Charge
- As we go down a group, the atomic number (Z) increases.
- This means more protons are present in the nucleus, resulting in a greater nuclear charge.
- Normally, an increase in nuclear charge would pull the electrons closer to the nucleus, reducing the atomic size.
- Addition of New Electron Shells (Principal Quantum Levels)
- However, moving from one element to the next down the group also adds a new electron shell (new principal quantum number, n). This additional shell greatly increases the distance between the outermost electrons and the nucleus.
- Even though the number of electrons in the outermost shell remains the same, the distance of this shell from the nucleus increases with each step down the group.
- Increase Screening (shielding) effect
- Increase Screening (shielding) effect of inner electrons reduces the attraction between the nucleus and the valence electrons.
- Dominance of Shell Addition Over Nuclear Attraction
- The increase in the size of the electron cloud due to the addition of shells and increase screening effect is more significant than the increased nuclear attraction.
- Therefore, the size of the atom increases as we move down the group.
Example.1 : Alkali Metals (Group 1)
| Element | Atomic Radius (pm) | Electronic Configuration | No. of Electron Shells |
|---|---|---|---|
| Li | 152 | 1s² 2s¹ | 2 |
| Na | 186 | 1s² 2s² 2p⁶ 3s¹ | 3 |
| K | 231 | 1s² 2s² 2p⁶ 3s² 3p⁶ 4s¹ | 4 |
| Rb | 244 | [Kr] 5s¹ | 5 |
| Cs | 262 | [Xe] 6s¹ | 6 |
Observation:
- Each step down the group from Li → Cs adds one new electron shell, causing a significant increase in atomic size despite the rise in nuclear charge.
Example.2 : Halogens (Group 17)
| Element | Atomic Radius (pm) | Electronic Configuration | No. of Electron Shells |
|---|---|---|---|
| F | 72 | 1s² 2s² 2p⁵ | 2 |
| Cl | 99 | 1s² 2s² 2p⁶ 3s² 3p⁵ | 3 |
| Br | 114 | [Ar] 4s² 3d¹⁰ 4p⁵ | 4 |
| I | 133 | [Kr] 5s² 4d¹⁰ 5p⁵ | 5 |
| At | 140 | [Xe] 6s² 4f¹⁴ 5d¹⁰ 6p⁵ | 6 |
Observation:
- Similar to alkali metals, new shells are added down the group, increasing the atomic size.
- The increase is less steep compared to Group 1 due to stronger nuclear attraction in nonmetals.
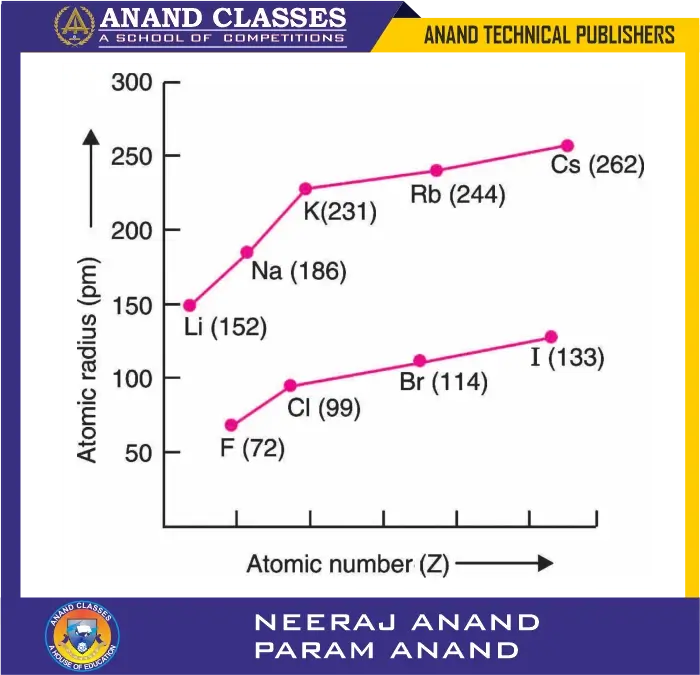
for alkali metals and halogens
Graphical Representation
When plotted, the atomic radius vs. element position in a group shows a steady upward trend from top to bottom. This confirms that addition of shells is the dominant factor in determining atomic size within a group.
FAQs on Variation of Atomic Radius in a Group
Q1. Why does atomic radius increase down a group?
A: As we move down a group, a new electron shell (principal quantum level) is added to the atom for each successive element. This increases the distance between the outermost electrons and the nucleus, making the atom larger despite the increase in nuclear charge.
Q2. Does increasing nuclear charge not decrease atomic size?
A: Normally, greater nuclear charge would pull electrons closer, reducing atomic size. However, in a group, the effect of additional electron shells outweighs the increased nuclear attraction, leading to a larger atomic radius.
Q3. How does the number of valence electrons change down a group?
A: The number of valence electrons remains the same for all elements in a group. For example, alkali metals always have one valence electron, and halogens always have seven, regardless of their position down the group.
Q4. Which group has the largest atomic radii — alkali metals or halogens?
A: Alkali metals (Group 1) have much larger atomic radii compared to halogens (Group 17) in the same period because alkali metals have fewer protons and weaker nuclear attraction for their valence electrons.
Q5. What is the trend in atomic radius among alkali metals?
A: It increases from lithium (Li) to caesium (Cs) because each element has an additional electron shell compared to the one above it.
Q6. What is the trend in atomic radius among halogens?
A: It increases from fluorine (F) to astatine (At) for the same reason — addition of extra electron shells down the group.
Q7. How does shielding effect relate to atomic radius in a group?
A: As more inner electron shells are added, the shielding effect increases, reducing the effective nuclear charge experienced by the valence electrons, which allows them to be farther from the nucleus, increasing atomic radius.
Do You Know? – Atomic Radius Trends
- Alkali metals (Group 1) have the largest atomic radii in their respective periods because they have only one valence electron and weak nuclear attraction.
- As we go down the group in alkali metals, the outermost electron gets farther from the nucleus and is more easily removed — this explains why reactivity increases down the group.
- Halogens (Group 17) have much smaller atomic radii than alkali metals in the same period because their nuclei have more protons pulling electrons closer.
- For halogens, atomic radius increases down the group, but reactivity decreases because the outer electrons are farther from the nucleus and harder to attract.
- The shielding effect increases with each new electron shell, reducing the effective pull of the nucleus on valence electrons.
- The addition of one electron shell per step down the group is the main reason for the increase in atomic size, even though nuclear charge also increases.
- Francium (Fr) is the alkali metal with the largest atomic radius, while Astatine (At) is the halogen with the largest atomic radius.
- Atomic radius is measured in picometres (pm) or angstroms (Å).
- The periodic table is arranged so that atomic radius decreases across a period but increases down a group — one of the key patterns in periodic properties.
For premium study materials specially designed for JEE, NEET, NDA, and CBSE/ICSE Classes, visit our official study material portal:
👉 https://anandclasses.net.in/
To enroll in our offline or online coaching programs, visit our coaching center website:
👉 https://anandclasses.co.in/
📞 Call us directly at: +91-94631-38669
💬 WhatsApp Us Instantly
Need quick assistance or want to inquire about classes and materials?
📲 Click below to chat instantly on WhatsApp:
👉 Chat on WhatsApp
🎥 Watch Video Lectures
Get access to high-quality video lessons, concept explainers, and revision tips by subscribing to our official YouTube channel:
👉 Neeraj Anand Classes – YouTube Channel


